
System overview
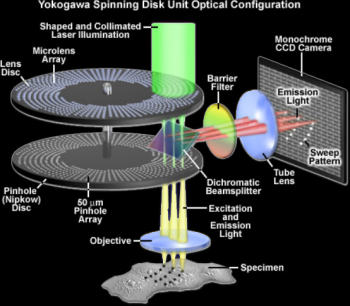 The Zeiss Cell Observer SD spinning disk confocal microscope is a fast imaging system which provides a trade-off between confocality, resolution and speed. It is an inverted microscope ideal for live cell applications which require fast acquisition speeds rather than high resolution images. The scanning unit achieves confocality by directing light through a spinning disk with many small pinholes. Images are then acquired with a sensitive EMCCD which allows for very small exposure times but is limited in resolution to 512×512 pixels. The stage is motorized and furthermore equipped with a piezo for Z displacement so fast 4D imaging is possible in multiple stage positions. The system is also equipped with a large size incubator for temperature control, a stage top incubator for CO2 supply and with Definite Focus.2 for hardware focus control during large tile or multi-position/time-lapse acquisitions (check tutorial videos below). You can also use the system as a widefield microscope, using fast-swicthing LEDs for illumination (Zeiss Colibri.2) and a sCMOS camera for acquisition.
The Zeiss Cell Observer SD spinning disk confocal microscope is a fast imaging system which provides a trade-off between confocality, resolution and speed. It is an inverted microscope ideal for live cell applications which require fast acquisition speeds rather than high resolution images. The scanning unit achieves confocality by directing light through a spinning disk with many small pinholes. Images are then acquired with a sensitive EMCCD which allows for very small exposure times but is limited in resolution to 512×512 pixels. The stage is motorized and furthermore equipped with a piezo for Z displacement so fast 4D imaging is possible in multiple stage positions. The system is also equipped with a large size incubator for temperature control, a stage top incubator for CO2 supply and with Definite Focus.2 for hardware focus control during large tile or multi-position/time-lapse acquisitions (check tutorial videos below). You can also use the system as a widefield microscope, using fast-swicthing LEDs for illumination (Zeiss Colibri.2) and a sCMOS camera for acquisition.
 Data files older than 1 month will be automatically deleted on this system, please copy your data to the GIMM server using the desktop link.
Data files older than 1 month will be automatically deleted on this system, please copy your data to the GIMM server using the desktop link.
System components
LASERs (confocal)
| LASER | Wavelength | Maximum Power |
| Violet | 405 nm | 50 mW |
| Blue | 488 nm | 100 mW |
| Yellow-Green | 561 nm | 75 mW |
| Red | 638 nm | 75 mW |
LEDs (widefield)
| Line | Wavelength |
| UV | 365 nm |
| B | 470 nm |
| G | 565 nm |
| R | 625 nm |
Filtersets (Widefield)
| Position | Filterset | Excitation | Dichroic | Emission | Reference |
| 1 | Green | 450-490 nm | 495 nm | 500-550 nm | FS38HE |
| 2 | Red | 538-562 nm | 570 nm | 570-640 nm | FS43HE |
| 3 | Blue | 335-383 nm | 395 nm | 420-470 nm | FS49 |
| 4 | Far Red | 625-655 nm | 660 nm | 665-715 nm | FS50 |
| 5 | QUAD | 357-393 nm | 400 nm | 407-412 nm | FS81HE |
| 473-496 nm | 503 nm | 511-528 nm |
| 545-562 nm | 570 nm | 577-602 nm |
| 620-642 nm | 650 nm | 658-704 nm |
| 6 | Empty |
Objectives
Upon request:
| Magnification | Model | Immersion | NA | WD (mm) | Reference |
| 10x | EC Plan-Neofluar | Air | 0.30 | 5.20 | 420340-9901-000 |
| 25x | LCI Plan-Neofluar Corr DIC | Oil/Glyc/W | 0.80 | 0.21 | 420852-9972-000 |
Emission Filters CSU X1
| Position | Setting | Transmission |
| 1 | 450 | 425-475 nm |
| 2 | TBP 526 601 688 | 510-542 nm |
| 586-616 nm |
| 663-713 nm |
| 3 | none | |
| 4 | 520 | 502-537 nm |
| 5 | 600 | 575-625 nm |
| 6 | 690 | 665-715 nm |
Cameras
Microscope Turn On Procedure
Turn on the HXP 200 fluorescent light source (if used)
Turn on the computer (if needed)
Login in to Windows with your Agendo credentials
Turn on the black socket outlet number 1 (back left corner of microscope table)

Wait for the Definite Focus controller to show App-Ver. +
OS-Image info

Turn on the black socket outlet number 2
Turn on the black socket outlet number 3
Start the ZEN Blue software
Microscope Turn Off Procedure
If there is another user for this microscope in the next hour:
Else:
Exit the ZEN software
Turn off the computer
Clean up immersion objectives (gently wipe with optic cleaning paper and EtOH embedded paper)
Turn off the black socket outlets by reverse order (3→2→1)
Turn off the HXP 200 fluorescent light source
Back to the Equipment page


 The Zeiss Cell Observer SD spinning disk confocal microscope is a fast imaging system which provides a trade-off between confocality, resolution and speed. It is an inverted microscope ideal for live cell applications which require fast acquisition speeds rather than high resolution images. The scanning unit achieves confocality by directing light through a spinning disk with many small pinholes. Images are then acquired with a sensitive EMCCD which allows for very small exposure times but is limited in resolution to 512×512 pixels. The stage is motorized and furthermore equipped with a piezo for Z displacement so fast 4D imaging is possible in multiple stage positions. The system is also equipped with a large size incubator for temperature control, a stage top incubator for CO2 supply and with Definite Focus.2 for hardware focus control during large tile or multi-position/time-lapse acquisitions (check tutorial videos below). You can also use the system as a widefield microscope, using fast-swicthing LEDs for illumination (Zeiss Colibri.2) and a sCMOS camera for acquisition.
The Zeiss Cell Observer SD spinning disk confocal microscope is a fast imaging system which provides a trade-off between confocality, resolution and speed. It is an inverted microscope ideal for live cell applications which require fast acquisition speeds rather than high resolution images. The scanning unit achieves confocality by directing light through a spinning disk with many small pinholes. Images are then acquired with a sensitive EMCCD which allows for very small exposure times but is limited in resolution to 512×512 pixels. The stage is motorized and furthermore equipped with a piezo for Z displacement so fast 4D imaging is possible in multiple stage positions. The system is also equipped with a large size incubator for temperature control, a stage top incubator for CO2 supply and with Definite Focus.2 for hardware focus control during large tile or multi-position/time-lapse acquisitions (check tutorial videos below). You can also use the system as a widefield microscope, using fast-swicthing LEDs for illumination (Zeiss Colibri.2) and a sCMOS camera for acquisition.
![]() Data files older than 1 month will be automatically deleted on this system, please copy your data to the GIMM server using the desktop link.
Data files older than 1 month will be automatically deleted on this system, please copy your data to the GIMM server using the desktop link.
 Zeiss Cell Observer SD Usage Statistics
Zeiss Cell Observer SD Usage Statistics
